The impact of enhancer RNAs (eRNAs) on promoter-proximal pausing by RNA polymerase II
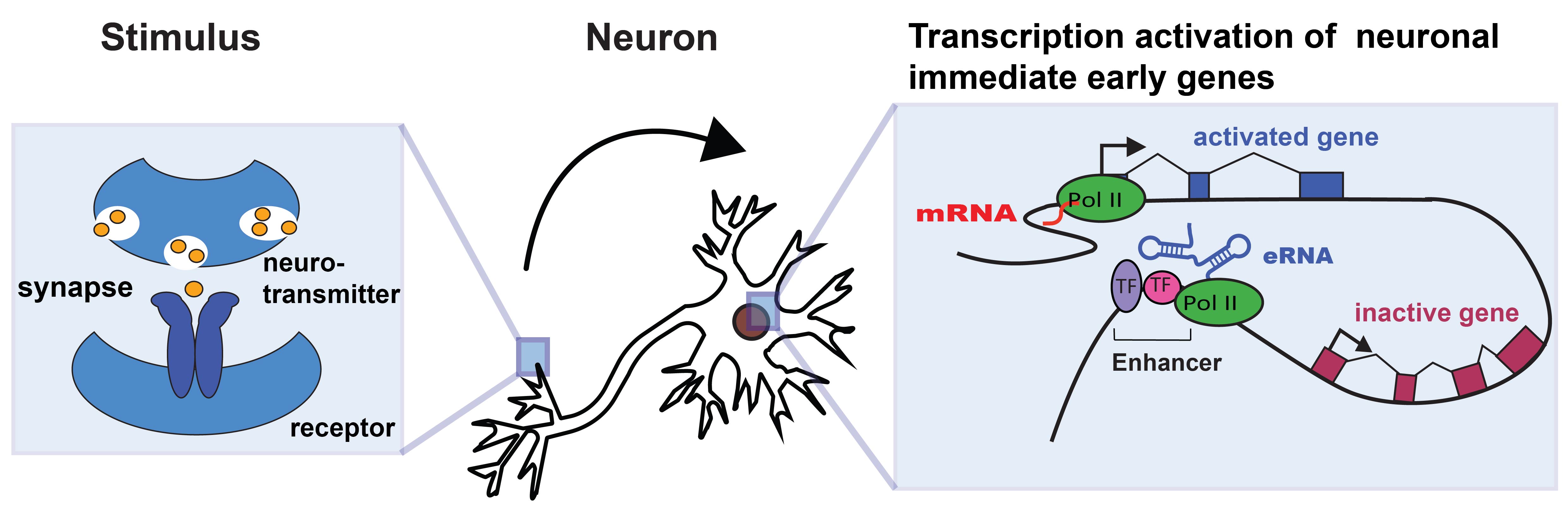
Stimulus-induced transcription activation of neuronal immediate early genes (IEGs)
In a second major focus of the lab, we ask how transcription by RNA polymerase II (Pol II) is modulated by enhancer RNAs (eRNAs). As their name indicates, eRNAs are long non-coding RNAs that originate from enhancer sequences. They are transcribed by Pol II and they were first identified at enhancer elements of immediate early genes (IEGs) and other rapidly induced genes across metazoans. Amongst others eRNAs were hypothesized to regulate promoter-proximal pausing of Pol II, an essential regulatory step in gene transcription in higher organisms. However, their mechanism of action remained unsolved. By combining a wide range of in vitro and in vivo techniques and in collaboration with T.-K. Kim from POSTECH in Korea, we significantly advanced our mechanistic understanding of eRNA function in neurons in a recently published manuscript (Gorbovytska et al. 2022).
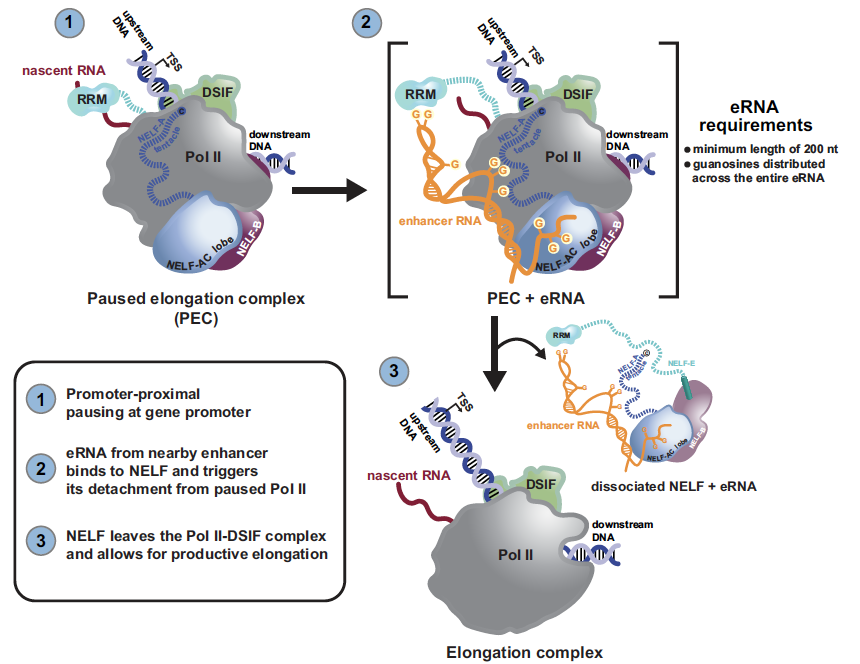
The figure above depicts a model of how multivalent, allosteric interactions between an eRNA and the paused elongation complex (PEC) stimulate Pol II pause release (Gorbovytska et al. 2022). Following the transcription of a nearby, activated enhancer, an eRNA molecule contacts the paused elongation complex (consisting of Pol II, DSIF, and NELF) at the target gene promoter. The eRNA interacts with NELF at multiple sites (positive patches on NELF-AC, the NELF-A and -E tentacles, and the NELF-E RRM domain). These multivalent, allosteric interactions collectively trigger NELF detachment from the PEC. The resulting elongation complex (consisting of Pol II and DSIF) then resumes transcription elongation. The length of the eRNA (>200) and unpaired guanosines within an eRNA are essential for its ability to detach NELF from the PEC.

A significant part of the projects in the Kuhn lab involve bioinformatics (mostly on piRNAs and eRNAs).

In vitro RNA production as well as the recombinant expression and purification of proteins (mostly in insect cells) constitutes a large part of our work on enhancer RNAs.
Collaborators:
Tae-Kyung Kim, Life Sciences @ POSTECH, Korea

